Iridium(III) chloride
 | |
| Names | |
|---|---|
| Other names
Iridium trichloride | |
| Identifiers | |
| (anhydrous), 14996-61-3 (hydrate) 10025-83-9 (anhydrous), 14996-61-3] (hydrate)] | |
| ChemSpider | 23837 |
| ECHA InfoCard | 100.030.028 |
| EC Number | 233-044-6 |
| PubChem | 82301 |
| UNII | 20278OEG45 |
| Properties | |
| IrCl3 | |
| Molar mass | 298.58 g/mol (anhydrous) |
| Appearance | dark green solid hygroscopic |
| Density | 5.30 g/cm3, solid |
| Melting point | 763 °C (1,405 °F; 1,036 K) (decomposes) |
| insoluble (anhydrous IrCl3), soluble (hydrated derivative) | |
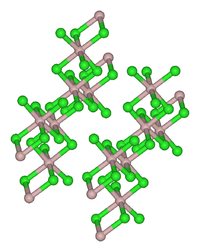
| Structure | |
| Monoclinic, mS16 | |
| C12/m1, No. 12 | |
| Thermochemistry | |
| Std enthalpy of formation (ΔfH |
-257 kJ/mol |
| Hazards | |
| EU classification (DSD) |
not listed |
| Flash point | non-flammable |
| Related compounds | |
| Other cations |
Rhodium(III) chloride |
| Related compounds |
Platinum(II) chloride |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Iridium(III) chloride is the inorganic compound with the formula IrCl3. The anhydrous compound is relatively rare, but the related hydrate is useful for preparing other iridium compounds. The anhydrous salt is a dark green crystalline solid. More commonly encountered is the trihydrate IrCl3(H2O)3.
Preparation
Iridium is separated from the other platinum group metals as crystalline ammonium hexachloroiridate, (NH4)2[IrCl6], which can be reduced to iridium metal in a stream of hydrogen. The spongy Ir thus produced reacts with chlorine at 300–400 °C to give iridium(III) chloride. Like the related rhodium compound, IrCl3 adopts the structure seen for aluminium chloride.[1]
Hydrated iridium trichloride is obtained by heating hydrated iridium(III) oxide with hydrochloric acid.
Uses
Industrially, most iridium complexes are generated from ammonium hexachloroiridate or the related chloroiridic acid (H2IrCl6) as these salts are the most common commercial forms of iridium chlorides.
Hydrated iridium(III) chloride is used in the laboratory for the preparation of other iridium compounds such as Vaska's complex, trans-[IrCl(CO)(PPh3)2].[2] Alkene complexes such as cyclooctadiene iridium chloride dimer[3][4] and chlorobis(cyclooctene)iridium dimer[4][3] can also be prepared by heating the trichloride with the appropriate alkene in water/alcohol mixtures.
Safety
Iridium(III) chloride is not listed under Annex I of Directive 67/548/EEC, but is usually classified as irritant, R36/37/38: Irritating to eyes, respiratory system and skin. It is listed in the inventory of the Toxic Substances Control Act (TSCA).
References
- ↑ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 0-08-037941-9.
- ↑ Vaska, L.; & DiLuzio, J. W. (1961) J. Am. Chem. Soc. 83:2784. Girolami, G.S.; Rauchfuss, T.B.; Angelici, R.J. (1999). Synthesis and Technique in Inorganic Chemistry (3rd Edn.). Sausalito:University Science Books.
- 1 2 Winkhaus, G.; & Singer, H. (1966). Iridium(I)-Olefinkomplexe. Chem. Ber. 99:3610–18.
- 1 2 Herde, J. L.; Lambert, J. C.; & Senoff, C. V. (1974). Cyclooctene and 1,5-Cyclooctadiene Complexes of Iridium(I). Inorg. Synth. 1974, volume 15, pages 18–20. doi:10.1002/9780470132463.ch5.